TobaccoIntelligence
COVID-19 news round-up
30th April 2020 - News analysis |
All TobaccoIntelligence’s COVID-19-related news briefings can now be found in one place here
All TobaccoIntelligence’s COVID-19-related news briefings can now be found in one place here
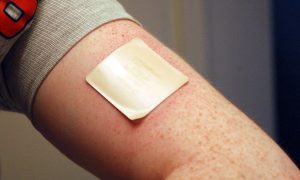
The French government is temporarily restricting sales of nicotine replacement therapy (NRT) products following research suggesting nicotine patches could be used in treatment for COVID-19
A group of non-profit and health organisations has called on Indonesian president Joko Widodo to take a stricter stance against tobacco products in the battle against COVID-19
Lawmakers in Kentucky have passed a new bill setting out taxes for tobacco products – but a proposal to increase tax on smokeless products was left out of the final wording
The US tobacco industry is resisting another attempt by the FDA to impose graphic health warnings, claiming in a lawsuit that mandatory warnings would violate their rights under the First Amendment
Legislative measures are on hold in half the states of the US due to the COVID-19 emergency, with most suspending activity until at least mid-April
The US Food and Drug Administration (FDA) has requested a 120-day extension to the deadline for premarket tobacco product applications (PMTAs) due to the challenges that the coronavirus outbreak is posing to companies
The Food Safety and Drug Administration in Uttar Pradesh, India’s largest state by population, has temporarily banned pan masala, a traditional oral product popular throughout India, to try to stem the spread of COVID-19
The Spanish government has announced plans to strengthen enforcement of existing tobacco regulations and to impose taxes on new products such as heated tobacco
Organisers of two major conferences and exhibitions on nicotine delivery are hoping the events can still go ahead despite the global coronavirus outbreak and a number of other event cancellations and postponements
Dutch health secretary Paul Blokhuis has called for heated tobacco and any future new tobacco product to be banned from public use in order to help achieve the target of a “smoke-free generation”
TobaccoIntelligence’s head of legal analysis, Pablo Cano Trilla, considers the options for regulation of tobacco-free snus (sometimes known as “white pouch”) under existing European Union law
Public Health England (PHE) has told TobaccoIntelligence it was never directly approached by Philip Morris International (PMI) over plans for a campaign to promote heat-not-burn and vaping products
For several decades, Australia’s political and medical establishments have taken a hard line against smoking; but do developments like heated tobacco call for a different approach? …
The US Food and Drug Administration (FDA) is to lose its power to regulate tobacco products if Congress passes president Donald Trump’s budget proposal for fiscal year 2021
The US FDA has taken a step closer to regulating nicotine levels in combustible cigarettes, but excluded vapour products from its proposals, even predicting a low-nicotine regime might encourage smokers toward electronic alternatives
As the US FDA gets to grips with its new focus on nicotine as a single marketplace, it is planning to take a fresh look at one of the longest-established non-combustible forms of the substance – nicotine replacement therapy
Scott Gottlieb, commissioner of the US FDA, appeared on Friday to go further than any federal official – or most politicians – previously had in supporting alternative nicotine products for harm reduction
Sign up to access our business and regulatory briefings and get the most updated news, insights and our expert analysis.